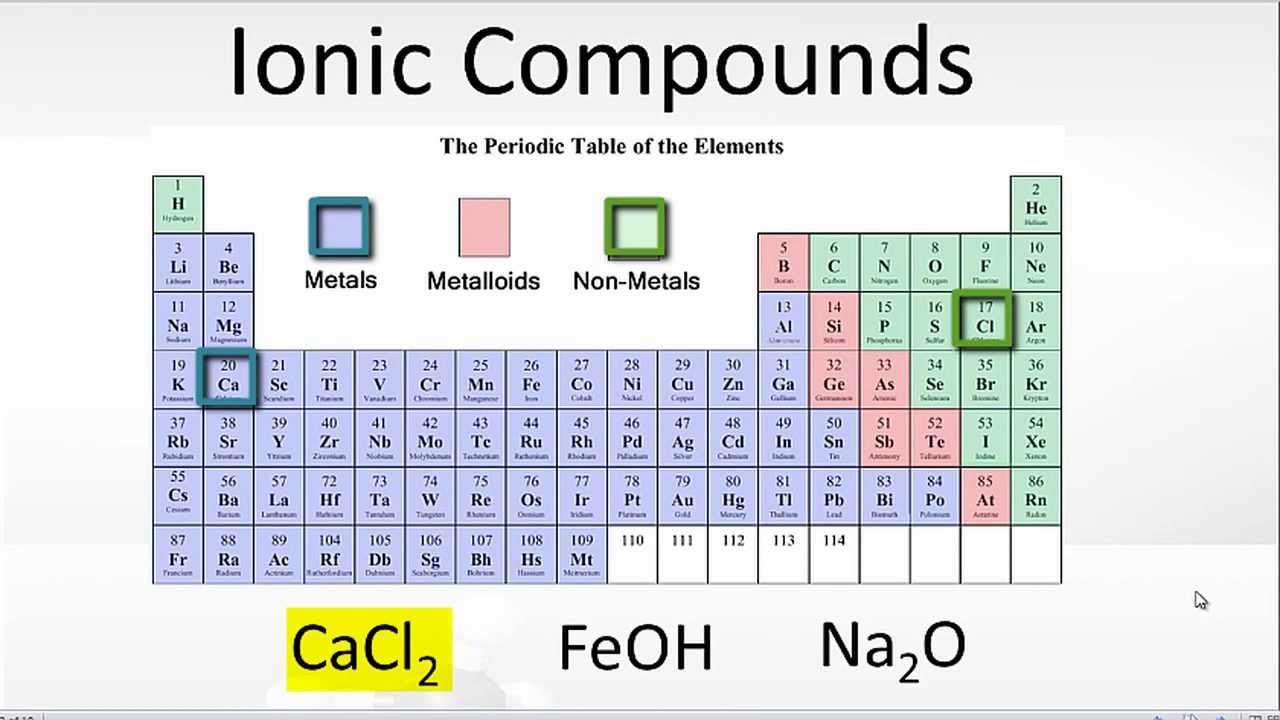
Are Ionic Bonds Formed Between Metals And Nonmetals . ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. By definition, a metal is relatively stable if it loses electrons to form. Binary ionic compounds are composed of just two elements: the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). A metal (which forms the cations). The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. the formation of ionic compounds. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons.
from studylibmoulders.z21.web.core.windows.net
A metal (which forms the cations). ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. Binary ionic compounds are composed of just two elements: the formation of ionic compounds. By definition, a metal is relatively stable if it loses electrons to form. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons.
How To Form Ionic Bonds
Are Ionic Bonds Formed Between Metals And Nonmetals A metal (which forms the cations). By definition, a metal is relatively stable if it loses electrons to form. A metal (which forms the cations). the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. Binary ionic compounds are composed of just two elements: the formation of ionic compounds. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity.
From www.slideserve.com
PPT Chapter 2 Lecture Notes—Essential Chemistry for Biology Biol 100 K. Marr Spring 2007 Are Ionic Bonds Formed Between Metals And Nonmetals By definition, a metal is relatively stable if it loses electrons to form. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). the formation of ionic compounds. the key difference between an ionic and covalent bond is that one atom. Are Ionic Bonds Formed Between Metals And Nonmetals.
From slideplayer.com
Chapter 2 Atoms, Molecules and Ions ppt download Are Ionic Bonds Formed Between Metals And Nonmetals The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. the formation of ionic compounds. ionic bonding results in compounds known. Are Ionic Bonds Formed Between Metals And Nonmetals.
From slideplayer.com
7.P.2A.4 Construct explanations for how compounds are classified as ionic (metal bonded to Are Ionic Bonds Formed Between Metals And Nonmetals A metal (which forms the cations). the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. Binary ionic compounds are composed. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.slideserve.com
PPT Chapter 1 Chemical Bonding PowerPoint Presentation, free download ID1482753 Are Ionic Bonds Formed Between Metals And Nonmetals By definition, a metal is relatively stable if it loses electrons to form. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. Are Ionic Bonds Formed Between Metals And Nonmetals.
From studynympholept.z21.web.core.windows.net
A Covalent Bond Forms Between Two Metals Are Ionic Bonds Formed Between Metals And Nonmetals the formation of ionic compounds. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. A metal (which forms the cations). Binary ionic compounds are composed of just two elements: By definition, a metal is relatively stable if it loses electrons to. Are Ionic Bonds Formed Between Metals And Nonmetals.
From dokumen.tips
(PPTX) Ionic Nomenclature. An ionic bond forms between metals and nonmetals. MetalsNonmetals Are Ionic Bonds Formed Between Metals And Nonmetals Binary ionic compounds are composed of just two elements: The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). By definition, a metal. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.youtube.com
Ionic bonds Reaction of metals & Nonmetals Metals and non metals Chemistry Khan Academy Are Ionic Bonds Formed Between Metals And Nonmetals the formation of ionic compounds. By definition, a metal is relatively stable if it loses electrons to form. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. A metal (which forms the cations). The ionic bond is normally formed between metals. Are Ionic Bonds Formed Between Metals And Nonmetals.
From slideplayer.com
Ionic and Covalent Compounds ppt download Are Ionic Bonds Formed Between Metals And Nonmetals Binary ionic compounds are composed of just two elements: By definition, a metal is relatively stable if it loses electrons to form. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. A metal (which forms the cations). the quick way of answering the question is looking. Are Ionic Bonds Formed Between Metals And Nonmetals.
From slideplayer.com
Atoms, Molecules, and Ions © 2009, PrenticeHall, Inc. Ionic Bonds Ionic compounds (such as NaCl Are Ionic Bonds Formed Between Metals And Nonmetals By definition, a metal is relatively stable if it loses electrons to form. the formation of ionic compounds. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). The ionic bond is normally formed between metals and nonmetals and the reason for. Are Ionic Bonds Formed Between Metals And Nonmetals.
From slideplayer.com
Chemical Bonds. ppt download Are Ionic Bonds Formed Between Metals And Nonmetals Binary ionic compounds are composed of just two elements: ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. the formation of ionic compounds. By definition, a metal is relatively stable if it loses electrons to form. The ionic bond is normally formed between metals and nonmetals. Are Ionic Bonds Formed Between Metals And Nonmetals.
From slideplayer.com
13.1 Electrons and Chemical Bonds ppt download Are Ionic Bonds Formed Between Metals And Nonmetals the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). A metal (which forms the cations). the formation of ionic compounds. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.slideserve.com
PPT Ionic Compounds Formula to Name PowerPoint Presentation, free download ID6776451 Are Ionic Bonds Formed Between Metals And Nonmetals Binary ionic compounds are composed of just two elements: A metal (which forms the cations). the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. the quick way of answering the question is looking at the periodic table and identifying which atoms. Are Ionic Bonds Formed Between Metals And Nonmetals.
From dokumen.tips
(PPT) Ionic & Covalent COVALENTIONIC A covalent bond is formed between two nonmetals that have Are Ionic Bonds Formed Between Metals And Nonmetals the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). By definition, a metal is relatively stable if it loses electrons to form. the formation of ionic compounds. Binary ionic compounds are composed of just two elements: the key difference between. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.expii.com
Ionic Bond — Formation & Compounds Expii Are Ionic Bonds Formed Between Metals And Nonmetals the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. By definition, a metal is relatively stable if it loses electrons. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.slideserve.com
PPT NonMetals PowerPoint Presentation, free download ID2409577 Are Ionic Bonds Formed Between Metals And Nonmetals the formation of ionic compounds. The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are nonmetals (h, cl). Binary ionic compounds are composed of just. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.slideserve.com
PPT Bonding PowerPoint Presentation, free download ID3645004 Are Ionic Bonds Formed Between Metals And Nonmetals Binary ionic compounds are composed of just two elements: the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. the quick way of answering the question is looking at the periodic table and identifying which atoms are metals (be) and which are. Are Ionic Bonds Formed Between Metals And Nonmetals.
From www.slideserve.com
PPT ELEMENTS PowerPoint Presentation, free download ID2738972 Are Ionic Bonds Formed Between Metals And Nonmetals Binary ionic compounds are composed of just two elements: The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. the quick way of answering the question is. Are Ionic Bonds Formed Between Metals And Nonmetals.
From studylibmoulders.z21.web.core.windows.net
How To Form Ionic Bonds Are Ionic Bonds Formed Between Metals And Nonmetals the formation of ionic compounds. the key difference between an ionic and covalent bond is that one atom essentially donates an electron to another atom in an ionic bond while electrons. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals. A metal (which forms the. Are Ionic Bonds Formed Between Metals And Nonmetals.